|
The basic idea is simple: spin the uranium atoms as if they were on a very fast merry-go-round. One initial proposal was to use a centrifuge. A method had to be developed that would sort atoms according to weight. The only difference among the uranium isotopes is their atomic weight. Chemical separation, developing a chemical reaction that will bind only uranium atoms, will separate out uranium atoms, but not distinguish among different isotopes of uranium. The difficulty is that all these forms of uranium, because they all have the same number of electrons, will have identical chemical behavior: they will bind in identical fashion to other atoms. The challenge is to separate this very small amount from the much larger bulk of other forms of uranium. Uranium 235, for example, makes up about 0.7 percent of the naturally occurring uranium on the earth. If radioactive, these usually are radioisotopes with very long half-lives. How can different isotopes of an element be produced? How can isotopes be produced-especially radioisotopes, which can serve many useful purposes? There are two basic methods: separation and synthesis. Iodine 131 thus became one of the earliest radioactive tracers. But, because it is radioactive, its presence can be detected. Because it behaves chemically as iodine, it travels throughout the body and localizes in the thyroid gland just like the stable form of iodine. 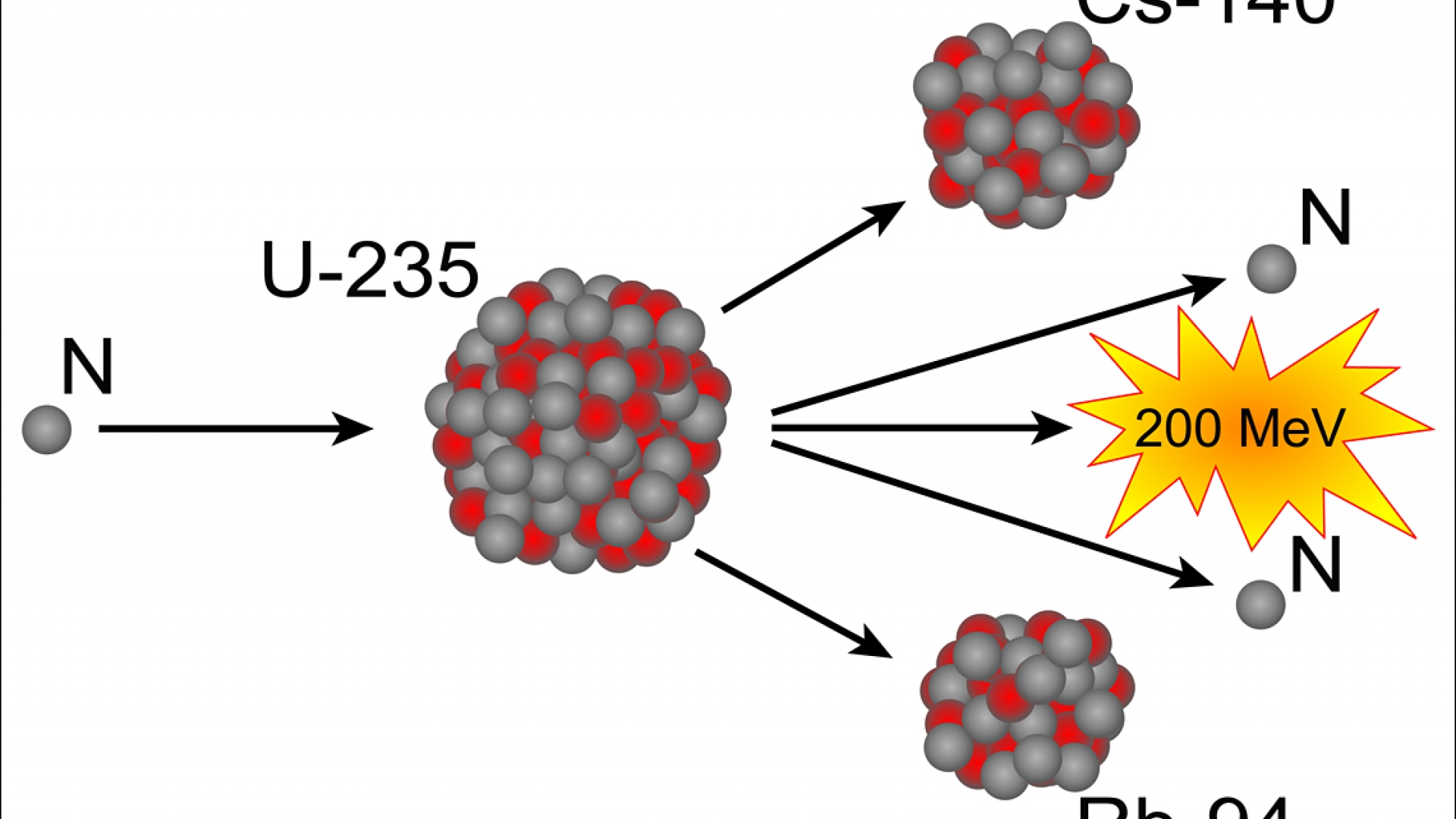
With "too many" neutrons in its nucleus, it is unstable and radioactive, with a half-life of eight days. A less stable form of iodine also has 53 protons (this is what makes it behave chemically as iodine) but four extra neutrons, for a total atomic weight of 131 (53 protons and 78 neutrons). Because its nucleus has the "correct" number of neutrons, it is stable and is not radioactive. The most common, stable form of iodine has an atomic number of 53 (protons) and an atomic weight of 127 (53 protons plus 74 neutrons). Iodine also is one of the earliest elements whose radioisotopes were used in what is now called nuclear medicine. The chemical behavior of U-235 is identical to all other forms of uranium, but its nucleus is less stable, giving it higher radioactivity and greater susceptibility to the chain reactions that power both atomic bombs and nuclear fission reactors.Īnother example is iodine, an element essential for health insufficient iodine in one's diet can lead to a goiter. Thus, it also has an atomic number of 92 (since the number of protons has not changed) but an atomic weight of 235 (92 protons plus only 143 neutrons). The isotope of uranium of greatest importance in atomic bombs, U-235, though, has three fewer neutrons. The most stable isotope of uranium, U-238, has an atomic number of 92 (protons) and an atomic weight of 238 (92 protons plus 146 neutrons). An isotope that is radioactive is called a radioisotope or radionuclide. But since they have different numbers of neutrons, these isotopes of the same element may have different radioactivity. Since they still have the same number of protons, all these isotopes of an element have identical chemical behavior. However, the isotopes of a single element vary in the number of neutrons in their nuclei. Radioisotopes: What Are They and How Are They Made? What are isotopes? The isotopes of an element are all the atoms that have in their nucleus the number of protons (atomic number) corresponding to the chemical behavior of that element. How Do Scientists Determine the Long-Term Risks from Radiation? How Do We Measure the Biological Effects of Internal Emitters? How Do We Measure the Biological Effects of External Radiation? 
What Are Atomic Number and Atomic Weight? New Ethical Questions for Medical Researchers The Aftermath of Hiroshima and Nagasaki: The Emergence of the Cold War Radiation Research Bureaucracy The Transformation in Government - Sponsored Research 
The Atomic Energy Commission and Postwar Biomedical Radiation Research The Manhattan Project: A New and Secret World of Human Experimentation In order to be neutral, an atom must have the same number of electrons and protons.Radioisotopes: What Are They and How Are They Made?ĭOE Openness: Human Radiation Experiments: Roadmap to the Projectīefore the Atomic Age: "Shadow Pictures," Radioisotopes, and the Beginnings of Human Radiation Experimentation 
If a neutral atom has 10 protons, it must have 10 electrons. If a neutral atom has 2 protons, it must have 2 electrons. If a neutral atom has 1 proton, it must have 1 electron. In other words, a neutral atom must have exactly one electron for every proton. This means that the negative charge on an electron perfectly balances the positive charge on the proton. Negative and positive charges of equal magnitude cancel each other out. \)) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly \(1\) in this unit system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
